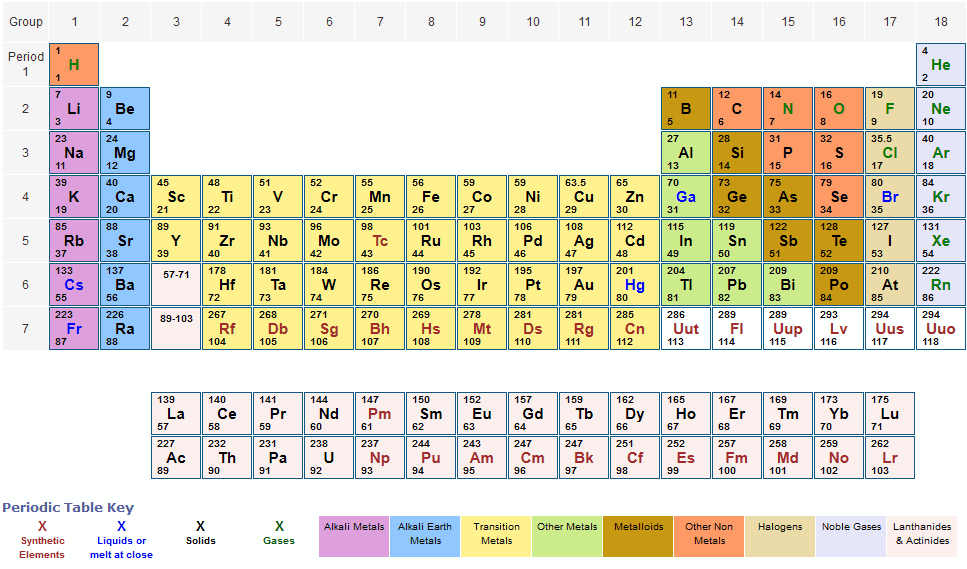

Table of Molar Masses: When you want to determine the molecular weight of different molecules, then put these weights in the molar mass equation to find the total weight of a molecule: Molar Mass. We created a timeline of the history of the periodic table. Periodic Table Elements: Enter Molecular Formula. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in W: Molar Mass (g/mol) W (Tungsten) 1 × 183.84 183.84. The Periodic Table has constantly been improved and developed over the past 200 years, but in 1869 Dimitri Mendeleev finished the first version of the periodic table as we know it today, by arranging the elements by atomic mass and leaving spaces open for the elements that were not yet discovered. The latest release of the Periodic Table (dated ) includes the most recent abridged standard atomic weight values released by the IUPAC Commission on Isotopic Abundances and Atomic Weights (), compiled as part of the 2021 Table of Standard Atomic Weights 2021. Who invented the Periodic Table of Elements? Immediately find any imaginable information about an element, calculate molar masses of any compounds you like, facilitate your overview with infographics and much more. The atomic weights used on this site come from NIST, the National Institute of Standards and Technology. The periodic table also gives us an idea of what the characteristics of an element might be and help us predict how an element might react based on in which group it is located. Using the chemical formula of the compound and the periodic table of elements, we can add up the atomic weights and calculate molecular weight of the substance. The Periodic Table of Elements can be used as an assisting tool in chemical calculations, when a specification of an element is needed it is easily found in the Periodic Table. How is the Periodic Table of Elements used? Finally, add together the total mass of each element to get the molar mass of O: 15.9994 g/mol 15.9994 g/mol. 1: Moles to Mass Conversion with Elements. According to the periodic table, 1 mol of U has a mass of 238.0 g, so the mass of 2 mol is twice that, or 476.0 g. The table lists all the elements that are currently known (118), in descending order of the number of protons that are present, in a single atom of the element. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in O: Molar Mass (g/mol) O (Oxygen) 1 × 15.9994 15.9994. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. The Periodic table of elements is a tool, developed by scientists over hundreds of years. List of all the elements and their properties:
